1
/
of
1
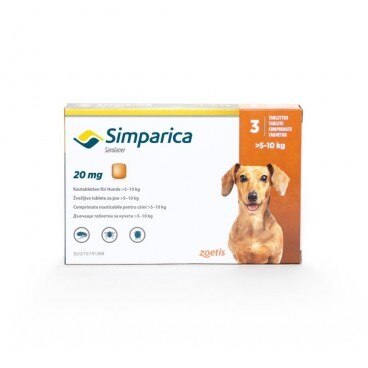
Simparica Dogs 3 tablets, pesticide for ticks and fleas and treatment of mange / per cani
Simparica Dogs 3 tablets, pesticide for ticks and fleas and treatment of mange / per cani
Regular price
€ 47.90 EUR
Regular price
Sale price
€ 47.90 EUR
Unit price
/
per
Shipping calculated at checkout.
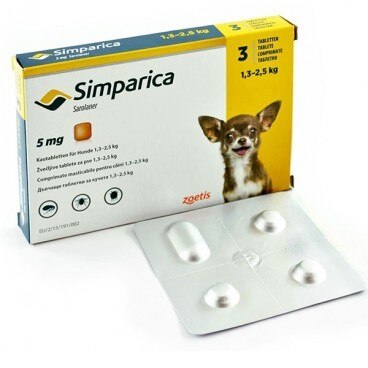
Simparica for Dogs 3 tablets are used in the treatment of mite infestation, flea infestation and sarcoptic mange in dogs. Qualitative and quantitative compositionEach tablet contains: chewable tablets 5 sarolaner simpara (mg)PHARMACEUTICAL FORM chewable tablets.Color mottled brown in a square shape with rounded edges.The number printed on one side refers to the strength of tablets (mg)CLINICSTarget species: Dogs.Indications for use, specifying the target species For the treatment of tick infestations (Dermacentor reticulatus, Ixodes hexagonus, Ixodes ricinus and Rhipicephalus sanguineus). The veterinary medicinal product has an immediate and persistent tick depleting activity up to at least 5 weeks.For the treatment of flea infestation (Ctenocephalides felis and Ctenocephalides canis). The veterinary medicinal product has activities to eliminate immediate and persistent fleas against new infestations for up to at least five weeks.The veterinary medicinal product can be used as part of a treatment strategy for flea allergy dermatitis (FAD).For the treatment of sarcoptic mange (caused by S. scabiei).Fleas and ticks, the host must be whipped and begins to feed in order to expose the active substance.Contraindications Do not use in case of hypersensitivity to the active substance or excipient.Special considerations for each target species Feeding parasites should begin to expose the sarolaner; therefore it cannot exclude the possibility of transmission of the disease through parasites.Special precautions for use in animals In the absence of available data, puppies less than 8 weeks of age and / or weighing less than 1.3 kg should not be treated with a veterinarian.Special precautions to be taken by the person administering the product to animals Wash your hands after handling the product.Accidental ingestion of the product can create adverse effects, such as transient excitatory neurological signs.To prevent children from accessing the product, remove the blister once tablet and only when necessary.Blisters must be returned to the box immediately and should not be stored in sight and reach of children.In case of accidental ingestion, seek medical advice immediately and show the package leaflet.Adverse reactions (frequency and seriousness) They don't know.Use during pregnancy, lactation or lay Safety of the product has not been established during pregnancy and lactation or in farm animals.Laboratory studies on rats and rabbits did not reveal the existence of teratogenic effects.Use only in accordance with the benefit / risk assessment by the responsible veterinarian.Interaction with other medicinal products and other forms of interaction They don't know.During clinical field studies, no interactions were observed between simpara chewable tablets for dogs and veterinary drugs commonly used in dogs.In laboratory safety studies, no interactions were observed when sarolaner was taken with milbemycin, moxidectin and pirantel pamoate (efficacy was not studied in these studies).Sarolaner engages intense plasma proteins and other proteins that can compete with intense conjugates, such as non-steroidal anti-inflammatory drugs (NSAIDs) and coumarin derivatives, warfarin. METHOD OF ADMINISTRATION For oral administration.The tablets can be taken with or without food.The veterinary medicinal product to be administered at a dose of 2-4 mg / kg body weight, according to the following table:Weight of the dog Quantity / day 1.3 - 2.5 kg One 5 mg tablet 2.5 - 5 kg One 10 mg tablet 5-10 kg One 20 mg tablet 10-20 kg One 40 mg tablet 20-40 kg One 80 mg tablet Chewable tablets are simpara and agreeable, quickly consumed by dogs when offered by the owner.If the tablets are not taken voluntarily in dogs, they can be administered with food or directly into the pet's mouthThe tablets must not be divided.treatment program: For optimal control of flea and tick infestations, the veterinary medicinal product continues to be administered at monthly intervals throughout the season. Need should be applied according to the local epidemiological situation repeated treatment.For the treatment of mange (caused by Sarcoptes scabiei var. Canis), one dose should be given at monthly intervals for two consecutive months.Overdose (symptoms, emergency procedures, antidotes), if necessary In the safety study, the medicinal product was administered orally to beagle dogs aged 8 months at doses of 0, 1, 3 and 5 times the maximum exposure dose.
Product features
Product features
Materials and care
Materials and care
Merchandising tips
Merchandising tips
Share